Recently a research group led by Professor Qun Tang from School of Pharmacy in Nanchang University published a research paper in the prestigious journal Redox Biology (Impact Factor: 11.9), a top-tier Category 1 journal according to the Chinese Academy of Sciences classification. The paper, titled “XPR1 downregulation inhibits hepatocellular carcinoma progression by suppressing serine metabolism,” identifies a novel regulatory mechanism in hepatocellular carcinoma (HCC) progression and reveals, for the first time, the functional role of the transmembrane phosphate transporter XPR1. Using multi-omics analysis, the group demonstrated that knockdown of XPR1 in HCC cells leads to significant suppression of serine metabolism. Follow-up in vitro and in vivo experiments further elucidated how XPR1 modulates serine metabolism and thereby influences HCC progression. Ziqiang Liao, a doctoral candidate from the Class of 2023 at the School of Pharmacy, Nanchang University, serves as the first author of the paper, with Professor Qun Tang acting as the corresponding author.

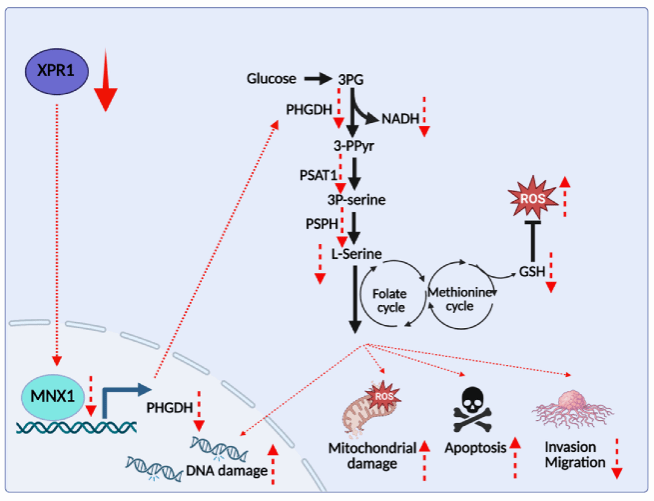
In detail, this study demonstrates for the first time that XPR1 is significantly upregulated in HCC tissues and that its expression correlates closely with patient prognosis. In cellular experiments, knockdown of XPR1 resulted in a marked reduction in serine levels within HCC cells, decreased protein expression of the key serine synthesis enzyme PHGDH, disruption of redox homeostasis, inhibition of cell progression-related functions, and significant mitochondrial fragmentation. The research identified MNX1 as a transcription factor of PHGDH. Restoring the expression of either MNX1 or PHGDH in XPR1-knockdown HCC cells effectively rescued redox capacity and reversed tumor progression-related functional impairments. Moreover, re-expression of PHGDH in these cells restored the tumorigenic capacity of HCC in animal models.
These results indicate that in HCC, XPR1 knockdown disrupts MNX1-mediated regulation of PHGDH expression, impairs serine metabolism, induces redox imbalance, and ultimately suppresses tumor progression. The findings highlight XPR1 as a promising potential therapeutic target for HCC, paving the way for subsequent investigations into the feasibility of developing targeted therapies. Additionally, this study uncovers a novel mechanistic role of XPR1—the sole phosphate efflux transporter in mammals—in tumor progression, particularly in modulating metabolic processes.
Original article link:
https://www.sciencedirect.com/science/article/pii/S2213231726000510
Author Profiles:
Ziqiang Liao, Doctoral candidate enrolled at Nanchang University in 2023.
Qun Tang, Full professor, Doctoral Supervisor. Distinguished Professor of the Ganjiang Scholars Program at Nanchang University. In recent years, he has conducted a series of studies in the fields of biomedical area, publishing over 30 SCI-indexed papers as a corresponding author. He has led more 10+ research projects, including those funded by the National Natural Science Foundation of China, Provincial key natural science funds, and enterprise collaborations. He was awarded the Second Prize of the Jiangxi Provincial Natural Science Award in 2023 (first-ranked contributor).
